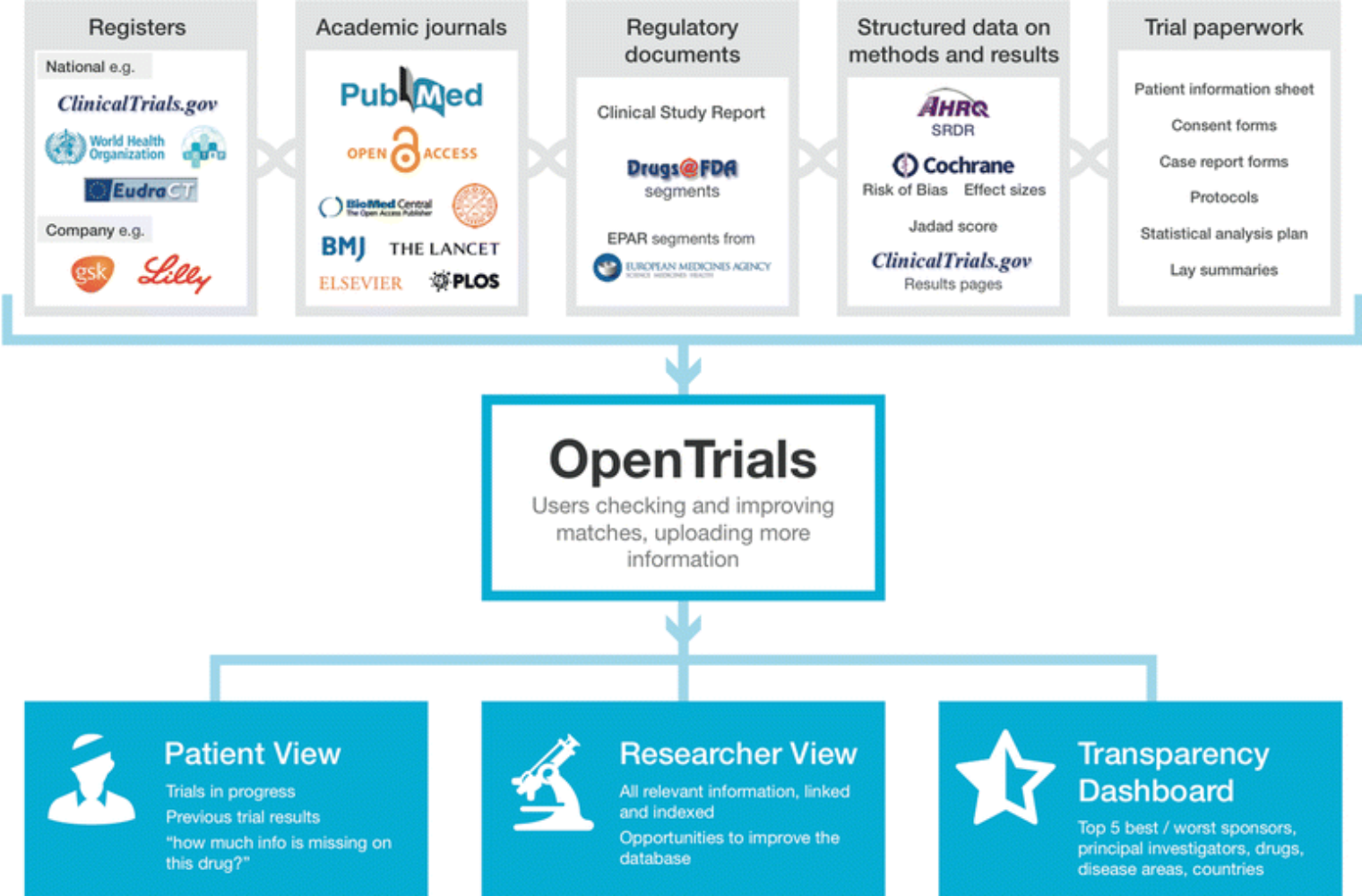
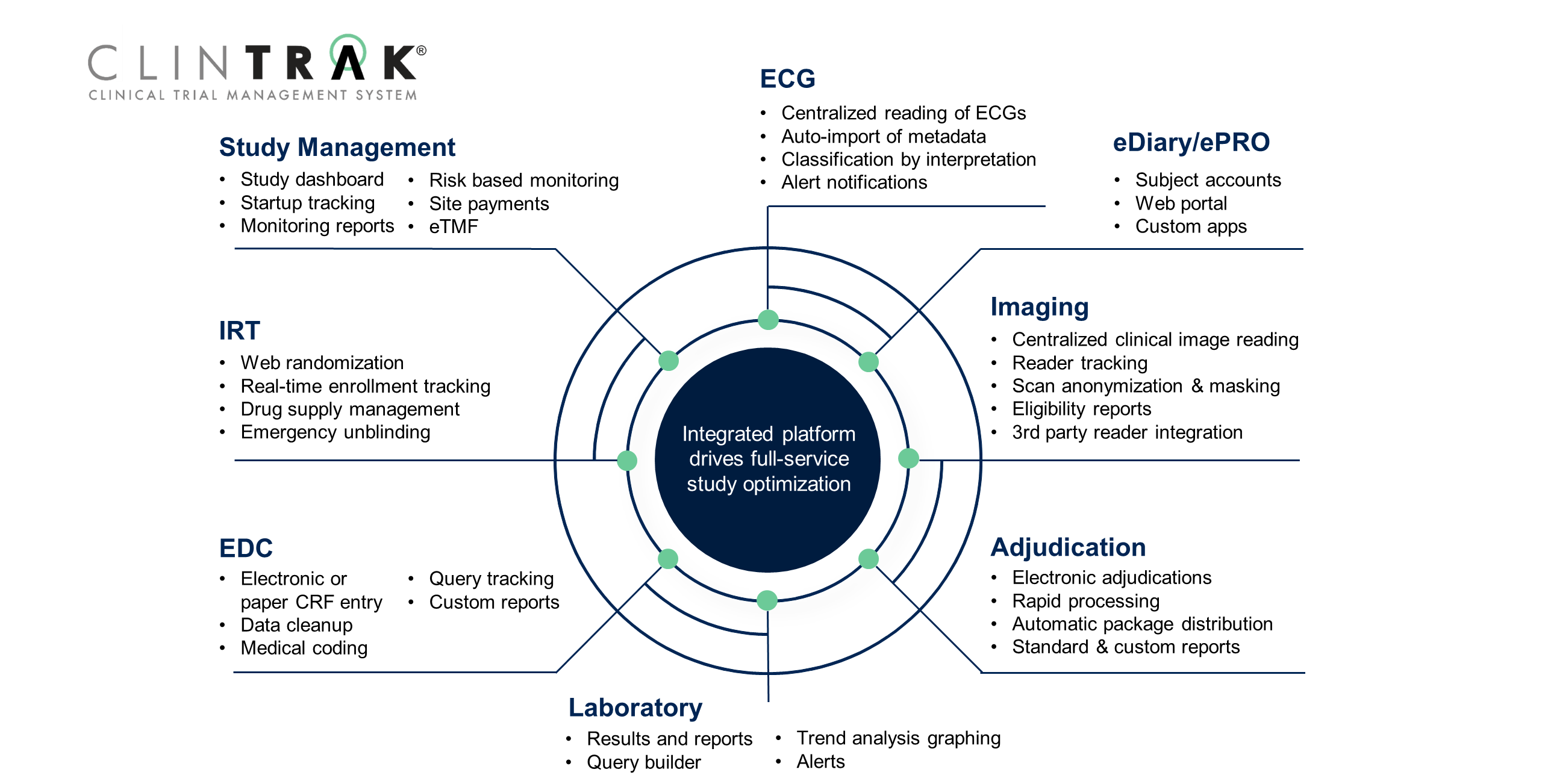
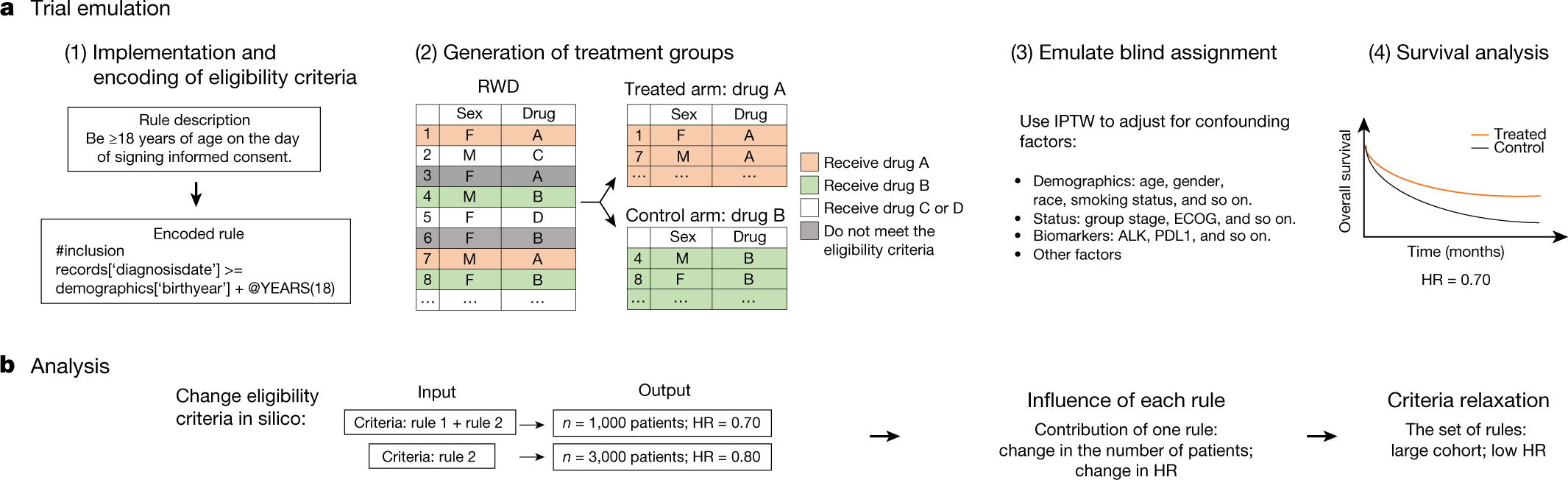
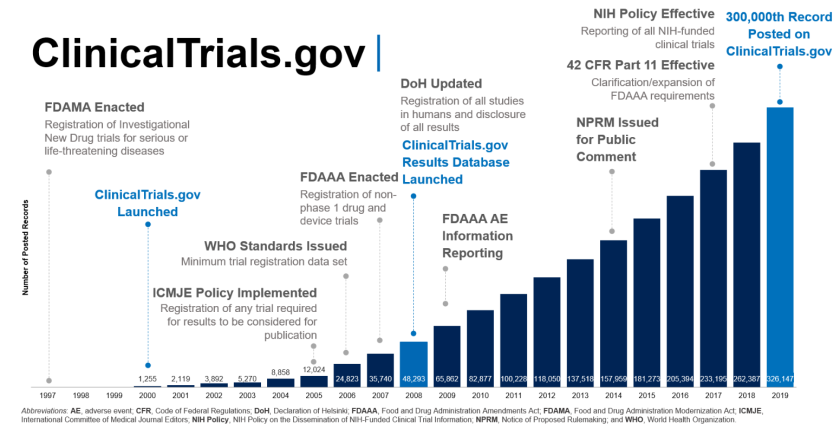
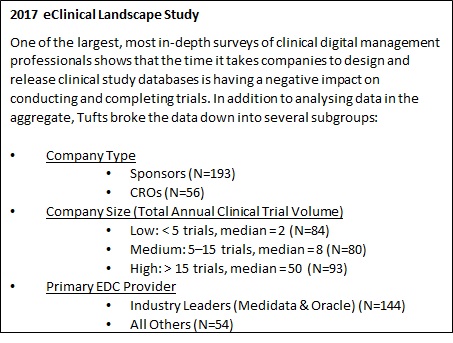
Clinical Trials - Grey Literature in the Health Sciences - Research Guides at University of Alabama - Birmingham

Clinical Trial Transparency: Ensuring Compliance with the New FDA Guidelines | BrackenData Clinical Trial Intelligence

A patient-level data meta-analysis of standard-of-care treatments from eight prostate cancer clinical trials | Scientific Data

Statistical controversies in clinical research: limitations of open-label studies assessing antiangiogenic therapies with regard to evaluation of vascular adverse drug events—a meta-analysis - Annals of Oncology
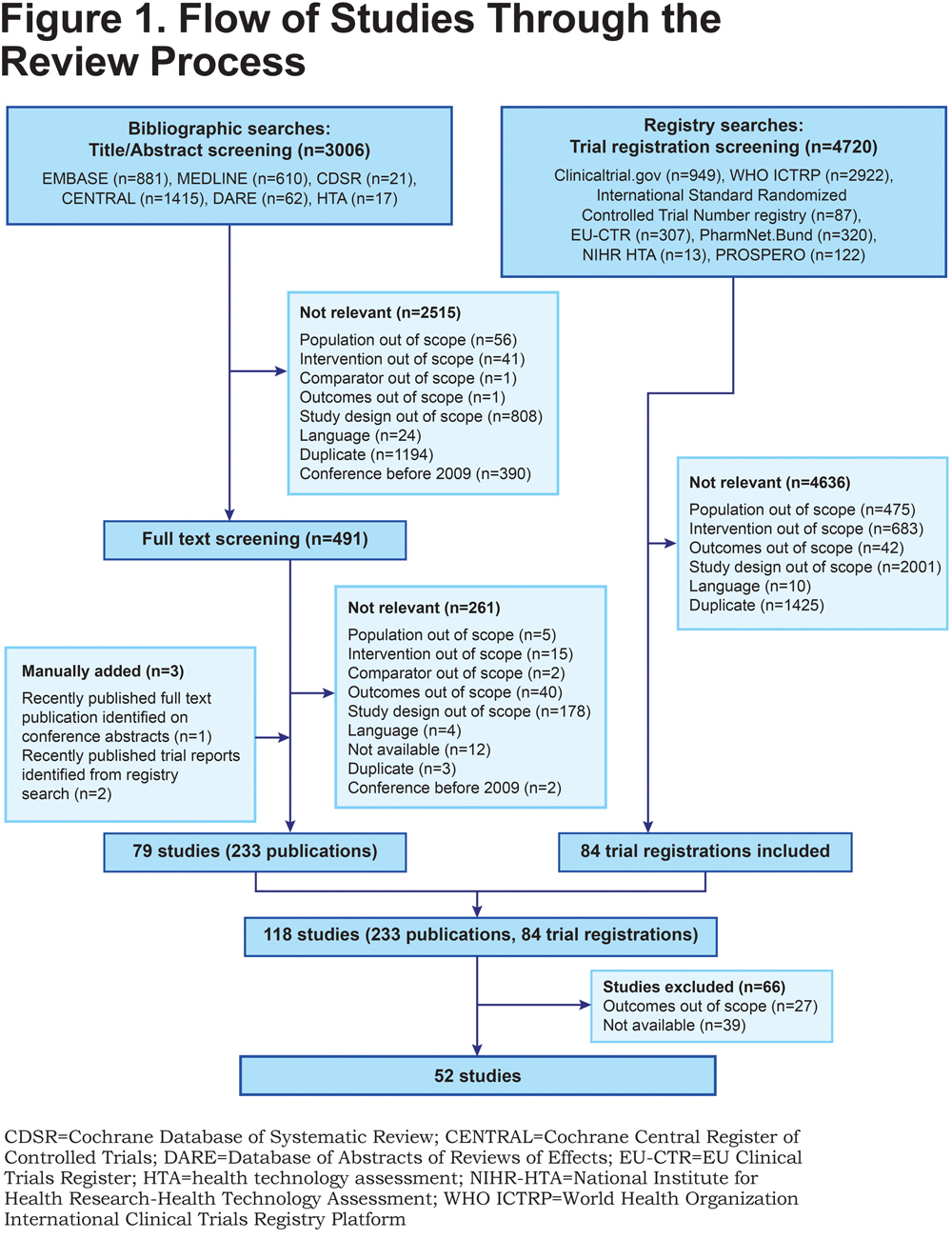
Relationship Between FEV1 and Patient-Reported Outcomes Changes: Results of a Meta-Analysis of Randomized Trials in Stable COPD > Journal of the COPD Foundation
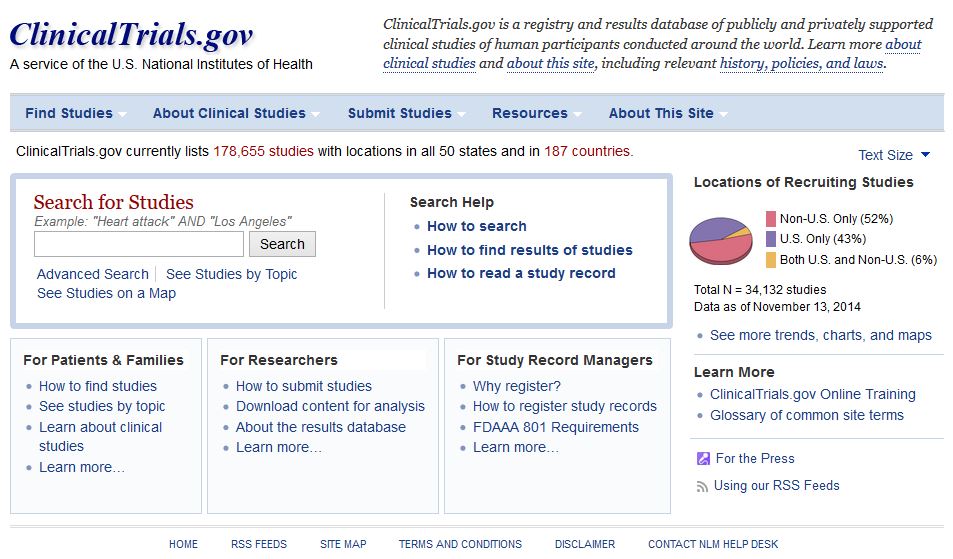
Investigator contact details should continue to be available after completion of clinical trials – DataSurg